Site sections
Editor's Choice:
- White spots on the nails, reasons for what to do, white spots on the nails and folk signs
- Available methods for rapidly increasing blood leukocytes
- Nail and skin fungus will not resist the coffee grounds
- Crocus furniture exhibition. Furniture exhibitions
- Owl tattoo on arm value
- The biggest members in the world
- Fractures of the phalanges of the foot photo
- What is “bad” and “good” cholesterol
- What to do if the skin around the nails dries
- The safest natural varnishes list
Advertising
| Lipid molecules are formed from. Lipid chemistry |
|
Lipids are the most important organic compounds that play a huge role in ensuring the functioning of the body. Without them, it is impossible to imagine more than one process occurring in our body. Lipids are part of cell membranes, create mechanical protection for organs, are precursors of biologically active substances - and this is not the whole list of functions. What are these connections? What are the classification and classes of lipids? Lipids are water insoluble substances. Most of them are the building blocks of cells, but these substances are also in free form. For transport of lipids in the blood requires special transport systems. Some compounds exist in a complex with albumin proteins. Most form water-soluble lipoproteins, which are composed of a lipid and apoprotein. Thus cholesterol and its esters, triglycerides and phospholipids are transported. Some of the lipids are involved in the formation of nanoparticles - liposomes. ClassificationSubstances of lipid nature are conveniently classified by structural features. Allocate simple and complex. These classes of lipids are hugely different. Simple differ in that they contain three standard chemical elements - it is oxygen, carbon and hydrogen. This group includes fatty acids, alcohols and aldehydes, as well as waxes and triglycerides. Complex substances have additional components - sulfur, phosphorus, nitrogen and others. They, in turn, are divided into polar and neutral. Among the polar - phospholipids containing a phosphoric acid residue. They also include sphingolipids, which are derivatives of amino alcohols. Neutral lipids are acylglycerides, sterol esters and ceramides. What is the difference between biochemistry? Simple lipids include only alcohol and fatty acids, and complex lipids correspond to their name. In addition to alcohol, they contain high molecular weight fats, as well as carbohydrates, residues of phosphoric acid. This is not the only one. Fat structureWhat is the difference between these substances? Biochemistry has studied the structure of their molecules. In saturated fats, all chemical bonds are filled with hydrogen molecules, while unsaturated ones are not. Due to this, their consistency is different - unsaturated, more liquid. Unsaturated fats can additionally be classified into monounsaturated and polyunsaturated. The first have only one vacancy for hydrogen, and the second - a few, such is their structure. Monounsaturated fats are found in oils such as olive, canola, and also in fish oil. Polyunsaturated enter the body with sunflower oil, oily fish, nuts. LipoproteinsAs mentioned above, lipids are insoluble in water and are transported by special transporters. The complex with apoproteins is called lipoprotein. The biochemistry of these substances differs in the density and size of molecules. Control of these substances is important for the timely prevention of atherosclerosis. Detailed analysis on When atherogenic fractions are detected, a special diet is prescribed, which depends on the individual characteristics. Its purpose is to reduce the flow harmful products - sausages, margarine, mayonnaise and so on. People with concomitant obesity should reduce their total calories per day. Role in the bodyWhat are the values of substances in the body? Lipids are involved in virtually all processes in the body, so their role is not limited to one function. Substances support livelihoods already at the molecular and cellular level. Structural functionRepresentatives of this group of substances are phospholipids, which are part of the bilayer of the cell membrane. Thus, lipids are the main structural substance of membranes. Their additional component is cholesterol, which is responsible for the property of fluidity. Biochemistry has studied that in membranes lipids are located in a special way. The heads of the molecules are hydrophobic and form a layer of the same name, and the tails are hydrophilic. The membrane consists of two layers of lipids, which are attracted by hydrophilic tails. Thus, a kind of barrier is being formed. The hydrophobic layer is of great importance, as it has the property of impermeability to polar compounds and ions. Thermal insulation and protectionFat cells accumulate in the subcutaneous tissue of warm-blooded animals, thereby reducing heat loss. Many organs have an additional layer that performs the function of mechanical protection. Energy function
 Lipids - backup energy sources. When they are oxidized, more energy is released than with the same process that occurs with carbohydrates. Biochemistry is simple - fat accumulates in the form of droplets localized in cells and, if necessary, is mobilized for energy needs. Lipids - backup energy sources. When they are oxidized, more energy is released than with the same process that occurs with carbohydrates. Biochemistry is simple - fat accumulates in the form of droplets localized in cells and, if necessary, is mobilized for energy needs. Regulatory functionThe value of lipids is also great for stabilizing all processes. This is due to the fact that lipids form the basis of important molecules. Thus, fat-soluble vitamins A, D, E and K are involved in metabolic and regenerative processes. In addition, vitamin E is responsible for the proper maturation of germ cells, and K ensures the production of plasma clotting factors, responsible for stopping bleeding and maintaining optimal rheology. Most hormones have a lipid structure (steroid). Also, these substances are part of eicosanoids. Hormones are involved in the regulation of metabolism, sexual function, regeneration. They are carried by blood, due to which they can act distally, that is, far from the place of formation. Eicosanoids, depending on the mechanism of formation, are divided into prostaglandins, thromboxanes and leukotrienes. All these substances are extremely important - they are involved in the formation of the inflammatory process, blood clotting, regulate arterial pressure and sexual function, as well as are direct participants in an allergic reaction. Lipids in the dietIt is important intake of lipids with food. Food products contain mainly triglycerides, which are the most important source of energy. Mandatory supply of saturated fatty acids, which are contained in meat, milk. Unsaturated is also found in vegetable oils, seeds, nuts. Cholesterol, found in animal foods - meat, eggs, butterHowever, you should not use them in excessive quantities. Meals should be balanced. The optimum ratio of proteins, fats and carbohydrates is 1: 1: 4. Adjustments can be made by a nutritionist individually for each case. The classification is based on the features of the molecules (structure). All these substances are involved in maintaining homeostasis, that is, constancy, in the body. Without them, existence is impossible. Based on natural lipids, the biochemistry of which has been carefully studied, were synthesized medicationsthat have been successfully applied in therapy. For example, glucocorticoids, used as anti-inflammatory, anti-allergic and immunosuppressive agents, are based on natural steroids. Currently, they help save the lives of patients, even in emergency situations. There are many such examples. Lipids are indispensable helpers of our body, without which it would not even exist. Lipid chemistry Lipids- These are organic substances that are poorly soluble or insoluble in water, but soluble in organic solvents; they are real or potential fatty acid esters. The content of lipids in the human body averages 10-20% of body weight. Lipids can be divided into two types: protoplasmic and reserve. Protoplasmic (constitutional) are part of all organs and tissues. They make up about 25% of all lipids in the body and almost remain at the same level throughout life. The reserve lipids are stored in the body and their number varies depending on various conditions. The biological significance of lipids in the body is great. So, they are found in the composition of all organs and tissues. The greatest amount (up to 90%) is contained in adipose tissue. In the brain, lipids make up half the body mass. Functions of lipids in the body: Ø Energy - along with carbohydrates are the main energy cell fuel. When burning 1 g of lipids, 38.9 kJ (or 9.3 kcal) is released. Ø Structural- lipids (phospholipids, glycolipids) together with proteins are part of biological membranes. Ø Protective - the function of mechanical protection, the role of which is performed by subcutaneous fatty tissue. Ø Thermoregulatory- implementation of this function is carried out due to two aspects: a) fat conducts heat poorly, therefore it is a heat insulator; b) when cooling the body to generate heat due to the release of energy lipids are consumed. Ø Regulatory- A number of hormones (sex, hormones of the adrenal cortex) are derivatives of lipids. Ø Lipids are a source of unsaturated higher fatty acids - vitamin F, one of the essential nutritional factors. Ø Fat is a source of endogenous water in the body. When oxidizing 100 g of lipids, 107 g of water is formed. Ø Lipids perform the function of natural solvents. They provide the absorption in the intestines of essential fatty acids and fat-soluble vitamins. Lipid classification Lipids
Washed neomylye
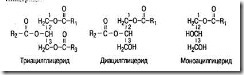
Simple Complicated Higher Higher Neutral Fats - Phospholipids alcohols hydrocarbons Waxes - glycolipids Sulfolipids Lipoproteins All lipids are divided into 2 groups: washed and untrimmed . Washingcalled the process of formation of salts of fatty acids by alkaline hydrolysis of fat. Soap- These are sodium or potassium salts of fatty acids. Sodium salts are solid soaps, and potassium salts are liquid. There are two classes of washed lipids: simple and complicated lipids. Simple lipids got their name because they consist only of atoms C, H and O. These include two groups of compounds: neutral fats and waxes. Simple lipids This group includes substances that are esters of alcohols and higher fatty acids. Of the alcohols in the composition of lipids are: glycerin, oleic alcohol and cyclic alcohol - cholesterol. Triacylglycerol (TAG) (triglycerides, neutral fats). They are esters of glycerin and three higher fatty acid molecules. TAG - the main components of apodocytes of adipose tissue, which is a depot of neutral fats in humans and animals. TAGs have the following structure: H 2 C - O - C = R NS - O - C = R 1 H 2 C - O - C = R 2 Since glycerin is a triatomic alcohol, fatty acids can form esterconnectionsin three places. Accordingly, in the tissues of the body are found monoacylglycerides, diacylglyceridesand triacylglycerides.
The carbon atoms in the glycerol molecule are numbered in accordance with the stereochemical nomenclature. There are many different types of triacylglycerides that differ in the nature of the three fatty acid residues attached to the glycerol by the ester bond. If in all three positions there are residues of the same fatty acid, then such triacylglycerides are called simple.In this case, their names are determined by the name of the corresponding fatty acid. Examples of simple triacylglycerides can be tristearoylglycerol (three stearic acid residues in the composition), tripalmitoylglycerol. Triacylglycerides, which contain residues of two or three different fatty acids, are called mixed The melting point of neutral fats (TAGs) depends on the fatty acid composition. It increases with increasing number and length of fatty acid components. For example, at 20 ° C, tristearin and tripalmitin are solids, and triolein and trilinelein are liquid. It should be noted that triacylglycerides are completely insoluble in water, since they are absent in their composition polar groups. As for diacyl and monoacylglycerides, they have by polaritydue to the presence of free hydroxyl groups. Therefore, they partially interact with water. Triacylglycerides are soluble in diethyl ether, benzene, chloroform. Most of the neutral fats in the body of animals contain mainly residues of palmitic, stearic, oleic and linoleic fatty acids. The composition of neutral fat from different tissues of the same organism can vary significantly. So, subcutaneous fathuman is richer in saturated fatty acids than liver fat, which contains more unsaturated fatty acids. The fats of butter and milk contain the highest amount of short-chain fatty acids. Fatty acid - These are aliphatic carboxylic acids. They serve as a kind of building blocks for most lipids. Currently, over 70 fatty acids have been isolated from living organisms. They can be divided into two groups: 1) saturated fatty acids and 2) unsaturated fatty acid. From saturated fatty acids in the body are more common palmitic, stearicand, less commonly, lignocerol, have 24 carbon atoms. Fatty acids containing 10 or less carbon atoms are rarely found in animal lipids. Of unsaturated fatty acids Acids consisting of 18 carbon atoms are most widely represented in the body. These include oleic (has one double bond), linoleic(two double bonds) linolenic(three double bonds) and arachidonic (has four double bonds) acids. Linoleic and linolenic in the body are not synthesized , and therefore they are among the indispensable factors of nutrition and should regularly come from food - vegetable oils, where they make up to 95%. In human fats, palmitic, myristic and lesser amounts of stearic acid prevail, and of unsaturated ones, oleic, linoleic and linolenic acids prevail. Physico-chemical properties of lipids are determined by the properties of their constituent fatty acids. Thus, saturated fatty acids have a high melting point and, accordingly, animal fats, consisting mainly of these acids, melt with more high temperature. Fats in which unsaturated acids (vegetable oils) predominate have a lower melting point. Fatty acid unsaturation significantly affects their properties. With an increase in the number of double bonds, the melting point of fatty acids decreases, their solubility increases in non-polar solvents and they react more easily than saturated ones. So, unsaturated acids can attach different atoms at the place of double bonds. In the body, oleic acid having a double bond adds two hydrogen atoms and turns into stearic acid. All unsaturated fatty acids found in nature are liquid at room temperature. Prostaglandids - These are derivatives of fatty acids with 20 carbon atoms, incorporating a cyclopentane ring. Prostaglandins are found in all mammalian tissues and have diverse biological effects. Currently, several groups of prostaglandins are known: A, B, E, F, I, D, H, G. Among them, prostaglandins F 2 and F 2α predominate, the precursor of which is arachidonic acid. In humans, all cells and tissues, with the exception of red blood cells, synthesize prostaglandins. The mechanism of action of prostaglandins on cells is not fully understood. The biological effect of prostaglandins in the body is as follows:
Prostaglandins are involved in the inflammatory process, increasing it in the inflammatory focus. Inhibitors of the formation of prostaglandins are acetylsalicylic acid and other salicylates. Aspirin inactivates an enzyme that catalyzes the conversion of arachidonic acid to prostaglandins. This explains the anti-inflammatory effect of aspirin. Waxes - These are esters of fatty acids and higher monohydric or dihydric alcohols. The number of carbon atoms in such alcohols ranges from 16 to 22. These are solid substances that mainly perform protective functions. The waxes are the so-called natural waxesi.e. those synthesized by living organisms (beeswax; lanolin - wax, which is part of the fat, covering wool; wax, covering the leaves of plants). Complex lipids The complex lipids class includes groups of compounds: phospholipids, glycolipids and sulfolipids, lipoproteins. Phospholipids - complex lipids containing phosphorus. In addition to phosphoric acid in their molecules are alcohols, fatty acids, nitrogenous bases and some other compounds. Phospholipids are important for the body: they form the basis of biological membranes, are found in large quantities in the nervous tissue (brain tissue is 60-70% composed of phospholipids), there are many of them in the liver and heart. Depending on the alcohol they contain, they are subdivided into glycerophospholipids and sphingophospholipids. The root "phospho" in these names indicates that the composition of all substances of these groups includes the residue of phosphoric acid. Glycerophospholipids.
The general structural formula of glycerophospholipids includes the alcohol residue — glycerin, whose hydroxyl groups on the first and second carbon atoms form ester bonds with fatty acids. The hydroxyl group at the third carbon atom forms an ester bond with residues of phosphoric acid. Usually, some nitrogen-containing substance (choline, serine, ethanolamine) is attached to the phosphoric acid residue. The general formula of glycerophospholipids is as follows: H 2 C - O - C = O NA - O - C = O H 2 C - O - P - O - R 3 The simplest glycerophospholipid is phosphatidic acid. In the tissues of the body, it is contained in small quantities, however, it is an important intermediate in the synthesis of triacylglycerides and phospholipids. Most widely represented in the cells of various tissues phosphatidylcholine (lecithin) and phosphatidylethanolamine (kefalin) They have the amino alcohols, choline and ethanolamine, attached to the phosphoric acid residue. These two glycerophospholipids are metabolically closely related to each other. They are the main lipid components of most biological membranes. There are other glycerophospholipids in the tissues. In phosphatidylserine, phosphoric acid is esterified with a hydroxyl group of serine, and in phosphatidylinositol, with a six-atomic alcohol, with inositol. Phosphatidyl inositol derivative - phosphatidyl inositol-4,5-bisphosphate is an important component of biological membranes. When stimulated by the corresponding hormone, it splits. Its cleavage products (diacylglyceride and ipozitol triphosphate) serve as hormone-acting intracellular messengers. Glycerophospholipids are metabolically very closely related lysophospholipids.Their composition contains only one fatty acid residue. An example is lysophosphatidylcholine, which plays an important role in the metabolism of phospholipids. Sphingophospholipids. They contain in its composition dihydric unsaturated alcohol sphingosine. A representative of this group of compounds, widespread in the body is sphingomyelin. It consists of sphingosine, a fatty acid residue, a phosphoric acid residue, and choline. Sphingomyelin is found in the membranes of plant and animal cells. The nervous tissue, in particular the brain, is especially rich in sphingophospholipids. The role of phospholipids: Ø Participate in the formation of membranes. Ø Influence membrane functions - selective permeability, realization of external effects on the cell. Ø Form a hydrophilic membrane of lipoproteins, promoting the transport of hydrophobic lipids. A characteristic feature of phospholipids is their diphilicitythat is, the ability to dissolve both in the aquatic environment and in neutral lipids. This is due to the presence of pronounced polar properties of phospholipids. At pH 7.0, their phosphate group always carries a negative charge. The serine residue in the phosphatidylserine molecule contains alpha-amino and carboxyl groups. Therefore, at pH 7.0, the phosphatidylserine molecule has two negative and one positively charged groups and carries a total negative charge. At the same time, the fatty acid radicals in the phospholipid composition do not have an electrical charge in an aqueous medium and thus are a hydrophobic part of the phospholipid molecule. The presence of polarity due to the charge of the polar groups causes hydrophilicity. Therefore, at the interface between oil and water, phospholipids are arranged in such a way that the polar groups are in the aqueous phase, and the non-polar groups are in the oil phase. Due to this, in the aquatic environment they form a bimolecular layer, and when a certain critical concentration is reached, micelles. The participation of phospholipids in the construction of biological membranes is based on this. The treatment of diphilic lipid in an aqueous medium with ultrasound leads to the formation of liposomes. Liposome - a closed lipid bilayer, inside of which is part of the aquatic environment. Liposomes are used in the clinic, cosmetology as a kind of container for the transfer of drugs, nutrients to certain organs and for the combined action on the skin. Glycolipids - These are sphingolipids containing carbohydrates. Glycolipids are widely distributed in tissues. They are especially rich in the myelin sheaths of the nerves. The composition of glycolipids includes alcohol - sphingosine. Glycolipids do not contain phosphoric acid. Their molecules have polar, hydrophilic carbohydrate groups (most often D-galactose). There are two groups of glycolipids: cerebrosides and gangliosides. Cerebrosides:the molecule contains an alcohol, sphingosine, linked by an ester bond to a fatty acid residue (nerve, cerebronic, lignoceric) - this complex is called ceramide. The carbohydrate portion of cerebroside is represented by D-galactose, which is attached to sphingosine. Fatty acids found in cerebrosides are unusual in that they contain 24 carbon atoms. More common nerve, cerebronand lignocericacid. The composition of cerebrosides of other tissues (except nervous tissue) instead of galactose may include glucose. Gangliosideshave a complex structure. The structure of the molecule in addition to sphingosine, includes an oligosaccharide containing residues of glucose and galactose, as well as one or more molecules sialic acids (derivatives of amino sugars). Sialic acid - these are amino sugar derivatives. N-acetylglucosamine and N-acetylneuramic acid are dominant in the gangliosides. Gangliosides are usually found on the outer surface of cell membranes, especially the nervous one. Cerebroside and ganglioside distribution in the brain tissue was noted. If the white matter is dominated by cerebrosides, the gray matter is gangliosides. Sulfolipids - These are glycolipids containing a sulfuric acid residue. Sulfolipids (sulfatides) have a structure similar to cerebrosides, with the only difference that at the 3rd carbon atom of galactose, the residue of sulfuric acid is attached instead of the hydroxyl group. Lipoproteins - complexes of lipids with proteins. The structure is small spherical particles, the outer shell of which is formed by proteins (which allows them to move through the blood), and inner part - lipids and their derivatives. The main function of lipoproteins is lipid blood transport. Depending on the amount of protein and lipids, lipoproteins are divided into chylomicrons, very low density lipoproteins (VLDL) - pre-β-lipoproteins, low density lipoproteins (LDL) - β-lipoproteins, and high-density lipoproteins (HDL) –α-lipoproteins. Neodymy lipids
Neodymy lipids not hydrolyzed with alkali to release fatty acids. There are two main types of unsaponifiable lipids - higher alcoholsand higher hydrocarbons.
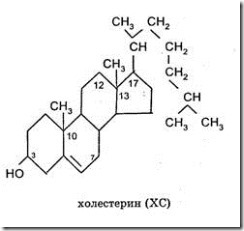
Higher alcohols To higher alcohols include cholesterol and fat soluble vitamins - A, D, E and F. Sterols- is a group of high molecular weight cyclic alcohols, which form esters with steric acids with fatty acids. The sterol representative is cholesterol(monatomic cyclic alcohol), first isolated from E. Conradi's gallstones in the 17th century. Cholesterol it is a derivative of cyclopentan perhydrophenanthrene containing three fused cyclohexane rings with which a cyclopentane ring is attached.
Cholesterol is a crystalline water-insoluble substance that can dissolve in organic solvents. Cholesterol is found in all cells of the body. Cholesterol is one of the main components of the plasma membrane and lipoproteins of the blood plasma, often in the body in esterified form (in the form of fatty acid esters) and serves as the starting compound for the synthesis of all steroids that function in the body (adrenal cortex hormones, sex hormones, vitamin D 3). No cholesterol found in plants. In the body, cholesterol performs important functions: Ø It is the precursor of many biologically important compounds: bile acids, steroid hormones, vitamin D, glucocorticoids and mineralocorticoids; Ø Included in the cell membrane; Ø Increases red blood cell resistance to hemolysis; Ø Serves as a kind of insulator for nerve cells, providing conduction of nerve impulses. Higher hydrocarbons Higher hydrocarbons are derivatives of isoprene. Among the lipid components that are found in cells and a relatively small amount are terpenes. Their molecules are built by combining several molecules of a five-carbon hydrocarbon - isoprene. Terpenes containing two isoprene groups are called monoterpenes, and those containing three are called sequiterpenes. In plants, a large number of mono-and sekviterpenov. Many of them give the plants their characteristic aroma and serve as the main components of the fragrant oils obtained from such plants. Carotenoids (precursors of vitamin A) belong to the group of higher terpenes. Natural rubber is a polyterpene.  Along with proteins and carbohydrates, lipids
are the main food elements that make up a significant part of the products power supply . The intake of lipids in the body with food has a significant impact on human health in general. Inadequate or excessive consumption of these substances can lead to the development of various pathologies. Along with proteins and carbohydrates, lipids
are the main food elements that make up a significant part of the products power supply . The intake of lipids in the body with food has a significant impact on human health in general. Inadequate or excessive consumption of these substances can lead to the development of various pathologies. Most people eat quite varied, and all the necessary lipids enter their bodies. It should be noted that some of these substances are synthesized cookies that partly compensates for their lack of food. However, there are irreplaceable lipids, or rather their components - polyunsaturated fatty acid . If they do not enter the body with food, over time this will inevitably lead to certain disorders. Most of the lipids in food are consumed by the body to produce energy. That is why when fasting man loses weight and weakens. Deprived of energy, the body begins to expend lipid reserves from subcutaneous fatty tissue. Thus, lipids play a very important role in healthy eating person However, for some diseases or disorders, their number should be strictly limited. Patients usually find out about this from the attending physician ( usually, gastroenterologist or nutritionist ). The energy value of lipids and their role in the dietThe energy value of any food is calculated in calories. The food product can be decomposed according to its composition into proteins, carbohydrates and lipids, which together make up the bulk. Each of these substances in the body disintegrates with the release of a certain amount of energy. Proteins and carbohydrates are easier to digest, but when 1 g of these substances decay, about 4 Kcal are released ( kilocalories) energy. Fats are more difficult to digest, but in the decay of 1 g about 9 Kcal is released. Thus, the energy value of lipids is highest.In terms of energy release, the greatest role is played triglycerides . Saturated acids that make up these substances are absorbed by the body by 30 - 40%. Monounsaturated and polyunsaturated fatty acids are completely absorbed by a healthy organism. A sufficient intake of lipids allows the use of carbohydrates and proteins for other purposes. Plant and animal lipidsAll lipids entering the body with food can be divided into substances of animal and vegetable origin. From a chemical point of view, the lipids that make up these two groups differ in their composition and structure. This is due to differences in the functioning of cells in plants and animals.Examples of sources of lipids of plant and animal origin What is the body's daily lipid requirement?Lipids are the main suppliers of energy to the body, but their excess can harm health. First of all, it concerns saturated fatty acids, most of which are deposited in the body and often lead to obesity . The optimal solution is to maintain the necessary proportions between proteins, fats and carbohydrates. The body should receive the amount of calories it spends during the day. That is why the lipid intake rates may vary.The following factors can influence the body's need for lipids:
Independently it is difficult for a person to make the necessary calculations and take into account all the factors for the selection of an optimal diet. To do this, it is better to consult a dietitian or food hygiene specialist. After a brief survey and clarification of the nature of the diet, they will be able to create an optimal daily diet, which the patient will adhere to in the future. They can also recommend specific foods that contain the necessary lipids. What foods contain mostly lipids ( milk, meat, etc.)?In one or another amount of lipids contained in almost all foods. However, in general, animal products are richer in these substances. In plants, the mass fraction of lipids is minimal, however, the fatty acids included in such lipids are most important for the body.The amount of lipids in a particular product is usually indicated on the product packaging in the “ the nutritional value". Most manufacturers are obliged to inform consumers about the mass fraction of proteins, carbohydrates and fats. In self-prepared food, the amount of lipids can be calculated using special tables for nutritionists, which list all the main products and dishes. Mass fraction of lipids in basic foods Are there essential lipids and what are their most important sources?The structural unit of lipids are fatty acids. Most of these acids can be synthesized by the body ( mostly liver cells) from other substances. However, there are a number of fatty acids that the body cannot produce on its own. Thus, lipids containing these acids are indispensable.Most of the essential lipids are found in foods of plant origin. These are monounsaturated and polyunsaturated fatty acids. The cells of the body cannot synthesize these compounds, since the metabolism in animals is very different from that in plants. Essential fatty acids and their main sources of nutrition What leads to a lack or excess of lipids in the diet?Both deficiency and excess lipids in the diet can seriously affect the health of the body. In this case, this is not about a single admission of a large amount of fat ( although it may have some consequences), and about the systematic abuse of fatty foods or prolonged fasting. At first, the body is fully capable of successfully adapting to a new diet. For example, with a lack of lipids in food, the most important substances for an organism will still be synthesized by their own cells, and energy needs will be covered by the breakdown of fat reserves. With an excess of lipids in the diet, a significant portion will not be absorbed into intestines and leave the body with fecal masses, and some of the lipids that enter the blood are transformed into adipose tissue. However, these adaptation mechanisms are temporary. In addition, they work well only in a healthy body.Possible effects of lipid imbalance in the diet Blood and plasma lipids A significant part of lipids is present in the blood in various forms. Most often these are compounds of lipids with other chemicals. For example, triglycerides and cholesterol are transported mainly in the form of lipoproteins. The level of various lipids in the blood can be determined using biochemical blood tests . This allows you to identify a number of violations and suspect the relevant pathology. A significant part of lipids is present in the blood in various forms. Most often these are compounds of lipids with other chemicals. For example, triglycerides and cholesterol are transported mainly in the form of lipoproteins. The level of various lipids in the blood can be determined using biochemical blood tests . This allows you to identify a number of violations and suspect the relevant pathology. TriglyceridesTriglycerides perform mainly energy function. They enter the body with food, are absorbed in the intestine and are carried throughout the body with blood in the form of various compounds. The normal content is the level of 0.41 - 1.8 mmol / l, but it can fluctuate significantly. For example, after eating large amounts of fatty foods, the level of triglycerides in the blood can increase by 2 to 3 times.Free fatty acidsFree fatty acids enter the bloodstream as a result of the breakdown of triglycerides. Normally, they are deposited in adipose tissue. Modern studies have shown the relationship between the level of free fatty acids in the blood and some pathological processes. For example, in people with a high concentration of fatty acids ( on an empty stomach) worse produced insulin therefore the risk of development diabetes above. The normal content of fatty acids in the blood of an adult is 0.28 - 0.89 mmol / l. In children, the boundaries of the norm are wider ( up to 1.10 mmol / l).CholesterolCholesterol is one of the most important lipids in the human body. It is part of a variety of cellular components and other substances, affecting a variety of processes. An excess or lack of this substance or a violation of its absorption by the body can lead to the development of serious diseases.In humans, cholesterol performs the following functions:
Lipoproteins ( lipoproteins) and their fractions ( low density, high density, etc.)The term lipoproteins or lipoproteins refers to a group of complex protein compounds that transport lipids in the blood. Some lipoproteins are fixed in the cell membranes and perform a number of functions related to the metabolism in the cell.All blood lipoproteins are divided into several classes, each of which has its own characteristics. The main criterion for distinguishing lipoproteins is their density. According to this indicator, all these substances are divided into 5 groups. There are the following classes ( fractionsa) lipoproteins:
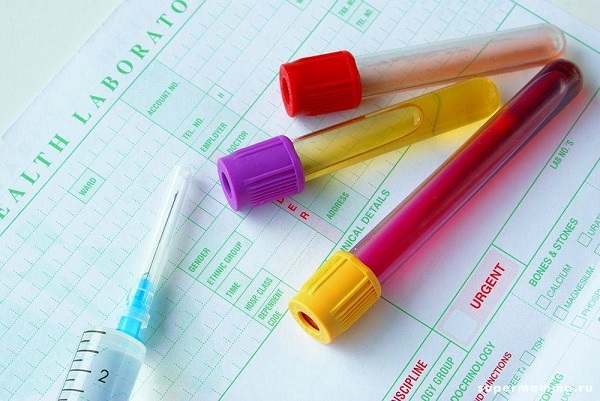
Lipid analysis Currently there are many laboratory tests that can be used to determine various lipids in the blood. Usually, venous blood is taken for this. The patient is sent for analysis by the attending physician. The most important lipids ( total cholesterol triglycerides) determined in the biochemical analysis of blood. If the patient needs a more detailed examination, the doctor indicates which lipids to be determined. The analysis itself usually lasts several hours. Most labs produce results the next day. Currently there are many laboratory tests that can be used to determine various lipids in the blood. Usually, venous blood is taken for this. The patient is sent for analysis by the attending physician. The most important lipids ( total cholesterol triglycerides) determined in the biochemical analysis of blood. If the patient needs a more detailed examination, the doctor indicates which lipids to be determined. The analysis itself usually lasts several hours. Most labs produce results the next day. What is a lipid profile?Lipidogram is a complex of laboratory blood tests aimed at finding out the level of lipids in the blood. This is the most useful research for patients with various disorders of lipid metabolism, as well as for patients with atherosclerosis. Some indicators included in the lipid profile are also determined in the biochemical analysis of blood, but in some cases this may not be enough to make an accurate diagnosis. Lipidogram prescribed by the doctor, based on the symptoms and complaints of the patient. This analysis is carried out by almost any biochemical laboratory.Lipidogram includes tests for the determination of the following blood lipids:
Before donating blood to a lipid profile, you should follow a few simple rules. They will help avoid significant fluctuations in blood lipids and make the results more reliable. Before passing the analysis, patients should consider the following recommendations:
Normal blood lipid levelsThe boundaries of the norm for all people are somewhat different. It depends on gender, age, the presence of chronic pathologies and a number of other indicators. However, there are certain limits, the excess of which clearly indicates the presence of problems. The table below shows the generally accepted boundaries of the norm for various blood lipids.The boundaries of the norm are relative, and the patient himself cannot always draw the right conclusions when interpreting the results of the analysis. When reviewing the results, the attending physician will necessarily take into account that during pregnancy the limits of the norm expand, as during fasting. Therefore, to panic with some deviations from the norm is not worth it. The final conclusion in any case must be made by the attending physician. Diseases associated with lipid metabolism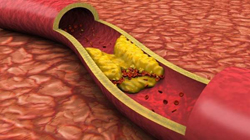 There are quite a few diseases that are to some extent associated with lipid metabolism in the body. Some of these pathologies cause an increase or decrease in various lipids in the blood, which is reflected in the analyzes. Other pathologies are a consequence of lipid imbalance. There are quite a few diseases that are to some extent associated with lipid metabolism in the body. Some of these pathologies cause an increase or decrease in various lipids in the blood, which is reflected in the analyzes. Other pathologies are a consequence of lipid imbalance. Disorders of lipid metabolism ( dyslipidemia)Excess or lack of lipids in the diet can lead to a variety of pathologies. In a healthy body that normally assimilates all incoming substances, this imbalance does not affect the metabolic processes. For example, excess lipids do not always lead to obesity. To do this, a person must also have a genetic predisposition, endocrine disorders, or he must lead a sedentary lifestyle. In other words, the amount of lipids in the diet in most cases is only one of the many factors influencing the appearance of pathology.An imbalance of lipids can lead to the following pathologies:
Causes of high and low lipid levelsThe most common cause of elevated lipids in the blood test are errors made during blood donation. Patients donate blood not on an empty stomach, because of which the lipid content does not have time to normalize, and the doctor may mistakenly suspect some problems. However, there are many pathologies that cause impaired blood lipids, regardless of nutrition.Pathological conditions associated with changes in the amount of lipids in the blood are called dyslipidemia. They are also divided into several types. If the blood levels of triglycerides are elevated, they speak of hypertriglyceridemia ( synonym - hyperlipemia). If the level of cholesterol rises, they talk about hypercholesterolemia. Also, all dyslipidemias by origin are divided into the following groups:
There are five main types of primary hyperlipoproteinemia ( elevated lipoprotein levels):
There are also secondary ( symptomatic) hyperlipoproteinemia in the following diseases:
Also elevated levels of lipoproteins in the blood can be observed when of pregnancy . This increase is usually negligible. With an increase in the level of lipids in 2 - 3 times higher than normal, it is necessary to consider the probability of pregnancy in combination with other pathologies that cause an increase in the level of lipids. What diseases of the digestive system are associated with lipid metabolism?A healthy digestive system is the key to good absorption of lipids and other nutrients. A significant imbalance of lipids in food over time can lead to the development of some pathologies. stomachOne of the most common problems incardiology is atherosclerosis. This disease is caused by the deposition of lipids in the vessels ( mainly in arteries). As a result of this process, the lumen of the vessel narrows and obstructs blood flow. Depending on which arteries are affected by atherosclerotic plaques, patients may experience various symptoms. Most characteristic high arterial pressure , coronary heart disease ( sometimes myocardial infarction), the appearance of aneurysms.Atherogenic lipids are those substances that lead to the development of atherosclerosis. It should be noted that the division of lipids into atherogenic and non-atherogenic is very conditional. In addition to the chemical nature of substances, many other factors contribute to the development of this disease. Atherogenic lipids often lead to the development of atherosclerosis in the following cases:
|
| Read: |
|---|
Popular:
Birch hanging or warty
|
New
- The program of intensive moisturizing of the skin on cosmetics bark
- What you need for acrylic powder
- What does owl mascot mean
- Analyzes for pancreatitis: what research should be done and what indicators show
- Owl - a talisman to attract money and good luck
- What bird screams at night with a kitten's voice?
- Cholesterol and stress
- Manicure at home
- Effective facial
- What is a man after a broken leg?